Virus Safety For Continuous Processing
By Mark Schofield, Ph.D.

The increased flexibility, efficiency, and quality that come with continuous processing are just some of the benefits driving the growth of this innovative alternative to batch manufacturing. From a regulatory perspective, the FDA has been encouraging industry efforts to move the industry to this modernized approach for the last several years. That is because the agency recognizes the advantages of continuous processing, where smaller equipment and facilities are leading to fewer steps and lower capital, operational, and environmental costs. The overall savings are then passed on to both the industry and, most importantly, the patients. However, among other regulatory questions surrounding this transition is the uncertainty around implementing an effective virus clearance strategy, which is intended to prevent various virus contaminations that could threaten patient safety.
Assessing Today’s Virus Clearance Methods
Virus safety for biologics is provided through a multifaceted approach. Raw materials and working cell banks are carefully chosen and screened to ensure they do not contain viruses. Tests are made during the process to confirm common virus contaminants are not present. Finally, the purification process has to be capable of robust virus removal. This includes multiple steps and orthogonal methods, such as chromatographic clearance, low pH virus inactivation, and virus filtration. Ensuring that these approaches can still be successful in the continuous processing context is a key part of enabling future adoption.
Chromatographic Clearance
Typically, virus clearance is provided by anion exchange chromatography, but clearance is often bolstered by the Protein A capture step. As anion exchange chromatography is typically performed in flow-through mode, its method of operation is not likely to change when transitioning to continuous mode. For bind and elute chromatography steps, such as Protein A, productivity and capacity can be improved by loading two or more columns in series. The first column can be overloaded to improve capacity while any product that breaks through can be captured on subsequent columns. A collaboration between Pall, the FDA, and Amgen is focused on determining what impact this change in loading has on the virus clearance through the Protein A step.1
The project was initiated with single-column experiments to understand virus clearance through the capture step. A bacteriophage was used to mimic a mammalian virus. The data from the experiments was then used to design experiments performed on a continuous chromatography skid. This enabled a comparison of virus clearance between single- and multi-column chromatography. The goal is to emulate the continuous process results with those achieved in batch processing and eventually confirm those findings with real mammalian viruses.
The Design of Experiments approach and subsequent Pareto chart in Figure 1 are used to identify the most important individual factors for virus clearance. Two parameters here are important for transition to continuous processing: the residence time, where there is 1 minute to mimic continuous or 4 minutes that is more typical for a batch process; and loading to percent breakthrough, where 5 percent is representative of some batch processes and loading to 60 percent breakthrough simulates what may occur in continuous processing. Neither of these factors is statistically significant, giving an initial indication that virus clearance may not be impacted by the transition to continuous processing. The results indicate three factors that have a major effect on virus clearance: the arginine in wash two, the sodium chloride in wash two, and wash CVs. These parameters are crossing to the right of the reference line for statistical significance at 95 percent confidence.
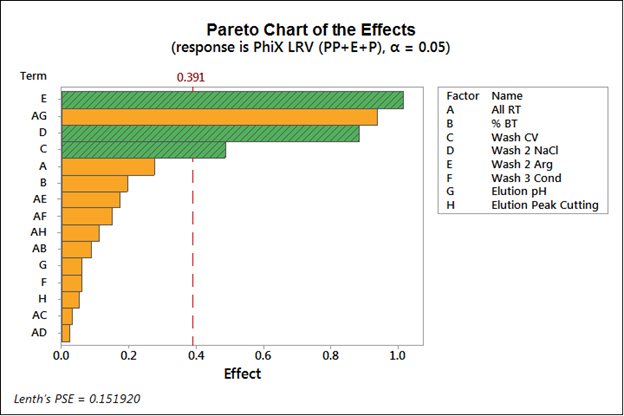
These parameters can then be used to look at two different continuous chromatography loading scenarios: a worst-case scenario where the lowest clearance is being achieved (just washing with three CVs, no additional salt, no arginine) or at the best-case scenario where the best clearance is being achieved (six CVs with an extra half molar salt and with 0.3 molar arginine). There is consistent virus clearance in continuous chromatography, with a large difference between the best- and worst-case scenarios. In the worst-case scenario, there is hardly any virus removal at all, but the best case shows two and a half logs. This data can be compared with results from the single-column experiments (all performed with 5 mL columns). In summary, there is a similar performance between the single- and multi-column scenarios and no indication there are any issues using this strategy for continuous processing, regardless of loading capacities or residence time.
Low pH Virus Inactivation
For batch processing, the low pH hold step, which is critical in virus inactivation, is most often performed manually. Many companies do not even have a pH probe in their pooling tank. The material in the tank is simply sampled and then measured on a benchtop pH probe, which can take a significant amount of time. Also, the product spends time near the inactivation pH, which is bad for the product but not low
enough to claim inactivation, so the process is more damaging for the product than it needs to be. To move to a continuous process, low pH virus inactivation must be automated. As the Protein A capture process may produce multiple eluates per hour and may be performed for several hours or even months, this step must also accommodate this change. As outlined in a recent article by Pall Biotech experts, different strategies have emerged as a result of these needs, including a continuous plug-flow reactor and packed column-based low pH hold to process and attenuate these elution volumes.2
However, the plug-flow approach presents a number of challenges, including the titration and how to manage the pH probes and residence time in the plug flow reactor. So, an alternative stirred tank approach was explored. In addition to its being identical to the well-known and well-understood low pH inactivation method commonly employed by the industry and accepted by regulatory authorities, the stirred tank approach’s advantages include:
- a reliable path to scale-down
- simplification of virus clearance validation studies, as the collection of the elution pool enables homogenization and eliminates any impact of protein concentration gradient on virus inactivation
- homogenization of product in preparation for subsequent purification steps
- segregation of batches (can trace and quarantine deviations)
- simple sampling strategy
Figure 2 below shows the flow path of the stirred tank system. Acid is added to achieve the inactivation pH. When it is reached, a 1-hour time is set. At the 1-hour point, base is added to neutralize the material in mixer one, and the pH is raised for the next process step. When the desired pH is reached, mixer one is emptied, and the material in mixer two is acidified while new material is being brought into mixer one. This is essentially a swing strategy where one tank is filling while the other tank processes and then switches over at the swing point.
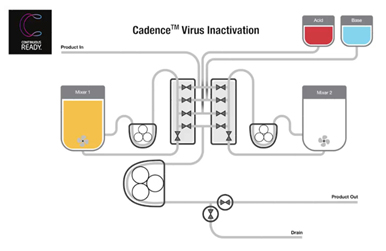
The system has a gamma-irradiated flow path and a central control unit designed for both continuous and batch operation that can operate fully automated semi-continuously to process a 2,000-liter fed batch bioreactor. Two key risks emerged while creating this system. The first was holdups that could contaminate inactivated product with non-inactivated material. The challenge with this is that bioprocesses are looking for five logs of removal. One way to eliminate this risk is having a flow pattern with no dead legs, which can be achieved by using an Aquasyn (Aquasyn LLC, NV) valve block. It minimizes holdup volume and creates a clear line of separation between incoming active elutions and inactivated elutions. A series of tests was performed to demonstrate the absence of dead legs. This involved mimicking the virus inactivation process using both bacteria and bacteriophage.
The other challenge is a lack of trust in pH probes. The team addressed this risk by screening pH probes using a process that mimicked the low pH process. The probe was first dipped in solution with a pH of 3.5 for 120 minutes. A robot arm then moved from that solution to one with a pH of 8 for 15 minutes and then moved into the air, where it was exposed for 15 minutes, then back to the solution with the pH of 3.5. This was done for 48 hours. A reference probe also used for the test solutions had its calibration checked throughout the experiments, which was then compared to the test probe. After periods of 24 and 48 hours, the same values were achieved for the test probe and the reference problem, with neither exhibiting any drifting throughout the experiments.
In the end, several benefits were achieved using this system. With the fluid entry at the base using a recirculation loop and low holdup valve block, there is negligible risk of contamination of virus inactivated product with non-inactivated material. There is also no requirement to have multiple tanks for one inactivation cycle. This fully automated operation with configurable predefined sequences eliminates operator error, runs continuously for long periods of time, and it is easy to configure. The multi-mixer processing allows for flexible operation in multiproduct facilities for both batch and continuous processes. It also makes Pall first to market with integrated chromatography and low pH virus inactivation solution for manufacturing scale, which integrates processing with unit operations running in parallel, leading to time and cost savings.
Virus Filtration
Traditionally, virus filtration is operated at a continuous and relatively high pressure for short periods of time. Some companies are looking at the potential to run semi-continuous, process intensified operation, which may run continuous upstream processes and chromatography processes but then hold the virus filter feed material until sufficient volume has been built up to allow the virus filtration and subsequent process steps to operate in a batch mode. This may be one of the common approaches to continuous in the short term; however, it does not represent where Pall believes the future of continuous processing is with a fully integrated continuous one-piece flow. This will require significant changes in the virus filter design space due to the continuous and low product stream flows.
A potential solution raised for continuous virus filtration is to run multiple small filters at equivalent flux to the batch process, resisting a change to the design space. This leads to far more risk and complexity, especially the risk of many more integrity tests at such a small scale. Pall recommends the use of fewer virus filters at lower flux to reduce filter switching complexity and improve risk. This makes the projected continuous virus filter design space very different from a batch process for the implementation of virus filtration in an ideal continuous process.
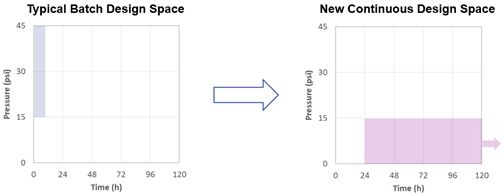
Figure 3 shows the typical batch and projected new continuous design spaces and the significant difference in operating pressure and operating time, allowing a virus filter to be run for several days at the lower, continuous flow expected during an optimal continuous process.
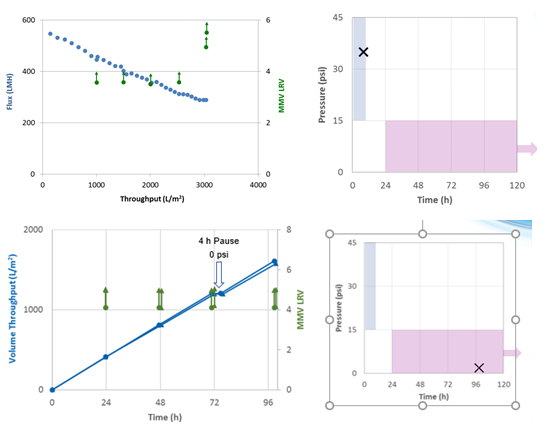
Because some virus filtration filters get breakthrough if the pressure is not high and constant, Pall Biotech designed the Pegasus™ Prime virus filter. Testing on the Pegasus Prime filters reveals the same virus clearance is achieved before and after a 30-minute pause during a batch process as well as before and after a 4-hour pause during a 96-hour continuous operation (Figure 4). No MMV was detected in any of the samples analyzed.
Summary
The overall goal of these experiments is to supply continuous solutions that will streamline current processes, in order to intensify processes. Changes to current chemistry or technologies are not being proposed. Drawing on the design space approach of quality by design and interacting earlier with regulators on these methods leads to a hybrid of new and traditional validation approaches that could facilitate the adoption of continuous processing and its benefits in biomanufacturing.
- Chiang, M.J., et. al. (2019, May 21). Biotechnology and Bioengineering. Validation and Optimization of Viral Clearance in a Downstream Continuous Chromatography Setting. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/31112283
- Schofield, Mark and Johnson, David M. (2018, September 1). Genetic Engineering and Biotechnology News. Supplement: Continuous Low-pH Virus Inactivation: Challenges and Practical Solutions. Vol. 38, No. 15. Retrieved from https://www.genengnews.com/magazine/324/supplement-continuous-low-ph-virus-inactivation-challenges-and-practical-solutions/
About the Author
 Mark Schofield earned his degrees in Scotland, he received his bachelor’s degree from the University of Edinburgh and his molecular biology Ph.D from the University of Dundee. For the last eight years he has been an employee of Pall life sciences focusing on continuous processing. Currently he holds the position of Senior R&D manager, his team works on chromatography solutions for next generation modalities.
Mark Schofield earned his degrees in Scotland, he received his bachelor’s degree from the University of Edinburgh and his molecular biology Ph.D from the University of Dundee. For the last eight years he has been an employee of Pall life sciences focusing on continuous processing. Currently he holds the position of Senior R&D manager, his team works on chromatography solutions for next generation modalities.
