Improve Operational Efficiency In Biomanufacturing With Comprehensive Automation Solutions

In the biopharmaceutical industry, process data is used in multiple ways throughout process development and the different production phases. However, equipment used are often connected only by process; that is, the bioprocess material passing between each of them. Often this connection is discontinuous, meaning equipment is controlled on an instrument-level, resulting in islands of automation. This white paper outlines how connectivity and comprehensive automation solution can facilitate technology transfer and scaling from process development to final manufacturing scale. Appropriate automation solutions also help maximize manufacturing efficiency and facility utilization.
Introduction
There is a variety of business models in the biopharmaceutical industry. Some companies focus on research, some focus on development, and yet others are centered on manufacturing. There are also companies that execute all these functions within their business. Effective business management is key in such complex operations as biopharmaceutical production. The ability to deliver safe, effective, life-saving therapeutics to patients relies on data and decisions driven by this data.
Data in the biopharmaceutical industry is used in a multitude of ways throughout the development and production phases of biotherapeutics (Fig 1). Decisions based on the information derived from this data vary by time and phase, from the scientific data associated with drug discovery to process data used in scale-up, manufacturing data used in the support of regulatory compliance and plant efficiency, and market data on target population size. In highly regulated industries such as the biopharmaceutical industry, data creates the foundation upon which quality and compliance are based. Automation is an essential element in the acquisition and management of data at each phase, from the supply chain to the boardroom. Comprehensive automation solutions allow capture and use of data in a way that enables production of biotherapeutics in a consistent manner compliant with regulatory authorities.
Whereas manufacturing typically focuses on one product with dedicated facilities, process development activities are routinely associated with multiple parallel drug candidates with development dispersed across several locations. At the development stage, processes are characterized and improved through experimentation and adjustments at bench scale. Increasing the process scale toward production volumes, the effect of scale progression on economic feasibility as well as product efficacy and quality is evaluated. Process versatility and experimental flexibility are important at this stage where process conditions are established, while making certain that the process continues to deliver product quality and yield comparable with previous scales. At the process development and scale-up stages, proprietary measurement and control systems developed by the instrument supplier are common. Each instrument or piece of equipment also has its own instrument-level controller.
Unlike in process development, where multiple parallel activities might be loosely or not at all integrated, an efficient manufacturing operation requires tight coordination of functional groups, raw materials, procedures, and the unit operations that make up the bioprocess train. Automation plays a central role in manufacturing coordination and control. Efficiency and facility utilization can be maximized with the appropriate automation architecture, especially by using an automation approach based on integration of the different unit operations.
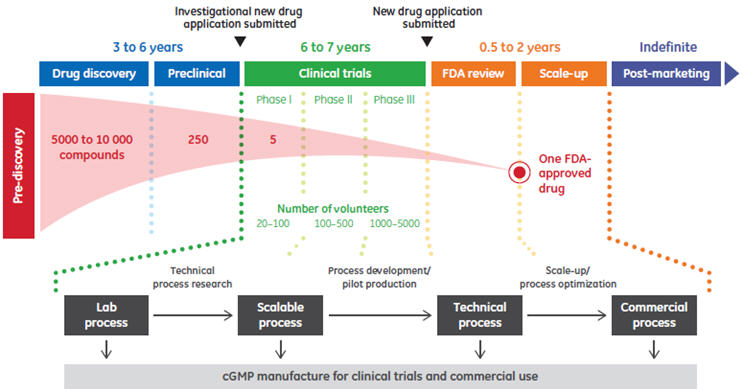
Fig 1. Overview of biopharmaceutical development and production.
Process development and optimization
During development, process equipment and systems meeting the application requirements need to be selected from a range of available products. Once selected, optimal process conditions are to be determined. For example, cell culture conditions for maximized productivity upstream or purification conditions for maximized yield and purity downstream need to be established. In addition, increasing demands from regulatory authorities for better process understanding is one of the cornerstones of the quality by design (QbD) initiative defined by the U.S. Food and Drug Administration (FDA). Supported by appropriate process analytical technologies, data generated at this stage contributes to greater process knowledge, resulting in a more efficient development process.
Design of experiments (DoE) is used to identify or screen input parameters that could affect the process output. Using a DoE approach, different process parameters can be varied simultaneously, allowing the effect of each parameter, individually as well as combined, to be studied. Although a sufficient amount of information about a process can be obtained in a minimum number of experiments using DoE, a large amount of data is created upon which informed decisions can be made.
Disjointed, or non-contiguous development activities constitute a challenge for companies trying to move fast in the competitive biopharmaceutical market. Although individual automated data collection and evaluation during process development contribute to reduce the time to market, automation solutions unifying separate development activities are preferred to accelerate commercialization.
Equipment and automation
Ahead of regulatory approval and the manufacturing phase, many potential therapeutic drug products might exist in a portfolio. Process development systems have the need for versatility, being able to handle as broad a process range as practical to manage diverse portfolios. The development stage is more tolerant of variability in process hardware and software (e.g., in process, data, operation, and documentation) than the manufacturing stage. Unlike in manufacturing, where space and equipment are dedicated to a given product, equipment used in process development might be preexisting from earlier development projects or new. If a piece of equipment used in development was originally designed with broad capabilities or can be easily modified, the capital investment can be extended.
Equipment used in process development is sourced from a variety of industry suppliers, having specific design features and attributes with little or no standardization between suppliers. These devices often include proprietary measurement and control subsystems. Proprietary, being defined as developed specifically by the respective supplier, may be further decomposed into two subcategories: (1) custom design, wherein custom electronics (circuit boards, housings, etc.) and custom software (often in a format using firmware for deployment) are used; and (2) industry standard hardware components and off-the-shelf software development tools. The latter makes specific reference to programmable logic controllers (PLC) or programmable automation controllers (PAC) and the software tools provided by PLC/PAC suppliers such as GE Fanuc™, Rockwell™/Allen- Bradley™, and Siemens™.
In automation terms, these proprietary measurement and control systems are at the instrument, equipment, or unit operation level. The control systems might even be different between unit operation or equipment sizes. In addition, each unit operation, whether bioreactor, filtration, or chromatography system, has its own instruments-level controller. Various pieces of equipment are connected only by process, that is, the bioprocess material passing between each of them. This connection is often discontinuous, with development activities physically separated between upstream and downstream teams (Fig 2). Besides meeting process performance objectives, data generated by the equipment must be available for consolidation with data from other systems. The consolidated data unifies disparate development activities, forming information from which to make appropriate decisions. Combining data for this purpose is left to the owner. Hence, systems must, at a minimum, have the ability to generate reports and allow export of data in a common format (e.g., tabular spreadsheet) and have interface compatibility with other systems, using a standard for open platform communication (OPC).
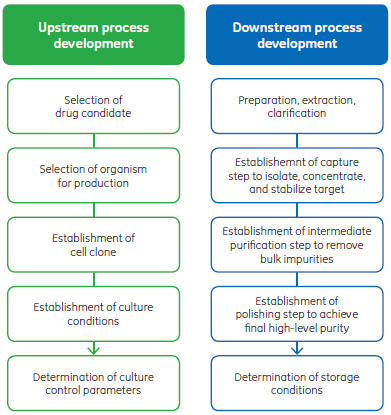
Fig 2. Process development activities are commonly dispersed between upstream and downstream teams. In addition, the parallel upstream and downstream process development activities can be separated into different departments, buildings, facilities, or even geographically.
Process scaling and transfer
The process at manufacturing scale needs to deliver equivalent purity and yield as the process during development. Biopharmaceutical process scale-up is generally complex, costly, and time-consuming. Upstream, scaling methodology is typically based on shear, tip speed, power, fluid velocity, among other considerations. Downstream, column packing, hydrodynamic pressure drop, and efficiency of liquid flow distribution need to be considered. Consistent geometry of included production vessels, such as cell culture bioreactors or chromatography columns, facilitates work at this stage. However, process parameters need to be easily transferred from one scale to the next. The use of modern system measurement and control software, allowing data to be exported in a spreadsheet-compatible format or exchanged with external systems using OPC, greatly facilitates transfer of the process from development to full manufacturing scale. Installed in a network, methods, or recipes, and run data can be easily shared among functional groups or securely stored on a common data base server.
Manufacturing automation
Biopharmaceutical production comprises preparation and storage of cell culture media and buffer solutions; multiple cell culture steps in different bioreactor technologies; protein purification steps using filtration and chromatography; and concentration, buffer exchange, and conditioning steps. All of these operations (through offline, manual, or semiautomated processes) create data sets that can facilitate batch release as well as process and application analyses. No longer acceptable are the non-contiguous activities often associated with process development. At the manufacturing stage, a highly integrated automation approach is a prerequisite for comprehensive data management and operational efficiency. With the appropriate automation architecture, process efficiency and facility utilization can be maximized. When combined with other enabling technologies such as single-use components and systems, a properly designed automation architecture can facilitate multi-product manufacturing. The S95 architectural model of Instrument Society of America (ISA) may be used as a guide in reviewing the manufacturing process, the automation, and the data flow (Table 1).
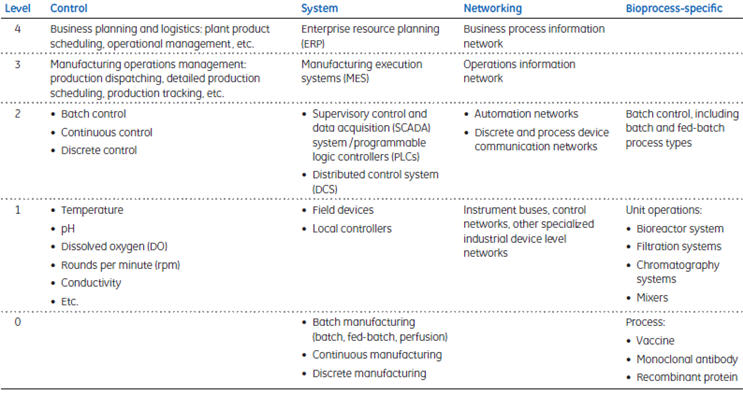
Table 1. S95 control hierarchy
ISA, an industry organization focused on instrumentation and control, has published numerous guidelines and standards for use in any industry where the publications can be put into practice. The ISA-95 standard includes three identified manufacturing types: discrete, batch, and continuous. Discrete manufacturing is associated with individual end-products such as a piece of equipment that can easily be individually identified by a serialized number. Batch, on the other hand, is associated with products made between specific process start and end times, and the product can be traced back to a specific production lot, or batch. Continuous manufacturing is exemplified by processes where the individual products cannot readily be identified as separate units. Oil and gas production is a typical example of continuous manufacturing.
The biopharmaceutical industry currently focuses on what is defined as batch manufacturing by ISA. Under batch, the three common bioprocess types are batch, fed-batch, and perfusion, each with its characteristic curves as exemplified in Figure 3. Perfusion represents the closest process type to continuous production, but has a definable duration, ranging from weeks to months.
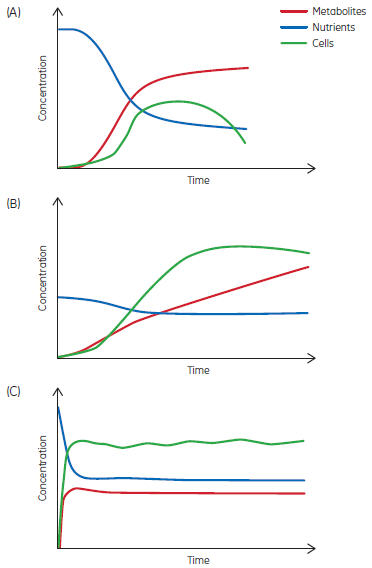
Fig 3. Examples of (A) batch, (B) fed-batch, and (C) continuous processes.
Level 0: the process
ISA-95 level 0 refers to the bioproduction process, for example, manufacturing of a vaccine, monoclonal antibody, or recombinant protein. Biomanufacturing today is based on organisms, either cell culture or microbial, with a finite growth period during which product is made in a batchwise manner. Regardless of how long the effective growth period is, at some point, the process is terminated, ending the batch. The automation architecture takes this production process as the basis for implementation at the next two levels (level 1 and 2).
Level 1: instrument-level control
ISA-95 level 1 relates to the individual unit operations and field devices for measurement and control of process parameters such as temperature, RPM, pH, DO, gas and liquid flow rates, weight, and pressure. Measurement field devices include probes and sensors, and their respective transmitters. Within the control field device category are such components as pneumatic valves, peristaltic pumps, and agitator drive motors.
Level 2: process automation
With plant-wide process automation, monitor and control of each individual instrument or unit operation (Level 1) can be coordinated to improve efficiency in operations. Connecting individual unit operations with an assortment of software tools simplifies intra-process communication as well as data transfer and archiving.
Components allowing device communication enable realtime transfer of process parameters between system control software and other plant-wide control systems, such as Rockwell, DeltaV™, Honeywell, or Siemens systems. There are two basic ways of achieving process automation based on integration of the different unit operations: (1) a plant-level SCADA system coupled to PLCs, or (2) a DCS. Implementation of SCADA/PLCs or DCS resides in ISA-95 level 2.
The SCADA/PLC scenario is a bottom-up, instrumentlevel approach, reflecting the equipment supply chain’s variability as it relates to equipment automation practiced by the numerous suppliers. Each unit operation potentially represents a unique island of automation, including dedicated measurements (temperature, pH, etc.), field devices (valves, pumps, motors), and local controllers. Typically, a set-point value entered in SCADA is sent to the PLC, which compares the measured value to the set-point and controls the process to match the set-point value. SCADA/PLC-based automation platforms are often used in control of batch or fed-batch processes.
The DCS is designed top-down, relying on distributed networks of measurement and control devices. The DCS approach offers a more comprehensive, highly integrated automation approach, taking into account an entire manufacturing process. In this environment, separate unit operations do not require instrument-level control, such as a PLC, eliminating islands of automation. Control is handled by the DCS. Only specific field measurements and control devices are needed at the equipment level. Typically, the DCS employs supplier-specific controllers (e.g., Emerson and Siemens) and both standard and proprietary protocols for communication, (e.g., Foundation fieldbus™, PROFIBUS™, and DeviceNet™). Measured process values are transmitted to the DCS across instrument and control networks. When the measured value reaches the set-point, the DCS instructs, for example, a valve to open or close until set-point level is matched. DCS-based automation platforms are commonly used in control of both batch and continuous processes. Plant-wide control is a native function of the DCS. Whereas SCADA/PLC-based platforms commonly execute recipes at the unit operation level, DCS platforms execute processwide recipes.
Level 3: manufacturing execution system (MES)
An MES is used to monitor and document processes from raw material to finished goods. The MES captures process data and outcomes in real time to be used in decision making, for example, in how to improve process output. An MES is used among cross-functional groups, for example, in production planning, detailed production scheduling, inventory, and production tracking.
An MES acts on ISA-95 level 3 to exchange data between the plant floor at level 2 and business planning and logistics functions at level 4. For example, data from level 2 is collected in a production tracking activity in the MES to generate a production performance report that is shared with the level 4 system. The information is interpreted at level 4 and the output can be used in the MES to aid in optimizing use of resources such as material, equipment, and personnel in production scheduling.
Level 4: enterprise resource planning (ERP)
The ERP system at ISA-95 level 4 offers an integrated overview of all production-related business processes. In the ERP system, data from different departments are collected, stored, and evaluated. This information is used in activities such as plant product scheduling and operational management. ERP systems facilitate sharing of information between different business functions and connection with external parties, for instance, with raw material inventory levels and their associated suppliers, and logistics companies responsible for finished product distribution.
Process intensification
Modern manufacturing concepts such as just-in-time (JIT), LEAN, and Six-Sigma are all applicable in biomanufacturing. Although biomanufacturing today has many requirements, some external (regulatory compliance, audit-ability, product efficacy, quality) and others internal (quality, reproducibility, cost of goods), maximal facility utilization is the goal. At the manufacturing level, facility utilization is essential. Getting the most product from the equipment and space is a competitive advantage. Whether achieved through more product per batch, more batches over time, or a combination of both, former, less productive ways of working will no longer survive.
Process intensification is commonly perceived as a promising path to reduce costs and improve throughput in the development and production of biopharmaceuticals. One way of achieving an intensified process is to transition from batch to a form of continuous processing. In a continuous process, product is moving through the process steps in an uninterrupted manner. As smaller equipment can be used and the need for intermediate hold-up tanks is minimized in a continuous process, the facility footprint can be reduced accordingly. As process steps are interconnected, the overall process time can also be reduced. Hence, productivity can be greatly increased in continuous processing compared with in batch processing.
Closed system operation is another way of decreasing number of process steps to reduce floor space requirement and shorten overall process time. Ready-to-use and singleuse products support closed system operation in that the products are ready for immediate use, omitting the need for separate product preparation and validation. The use of ready-to-use products reduces time for process setup and initiation. Adding single-use equipment to the process reduces changeover time between productions, as disposables require no cleaning and cleaning validation between batches.
A continuous process is maintained at steady state, meaning that quantities, such as feed input, temperatures, pressures, and output stream are kept within defined limits, simplifying surveillance and control. Continuous processes are easily integrated in process automation workflows supporting the process analytical technology (PAT) initiative defined by the FDA. Continuous processing using automated workflows also minimize contamination risk and opportunity for operator error by decreasing manual handling between process steps.
Unit operations on modular equipment platforms, in which the process hardware and the supplier’s preferred automation platform can be decoupled, simplifies integration into comprehensive automation solutions. An automation solution, spanning the complete upstream-downstream bioprocess train, under either the SCADA-PLC or DCS automation scenarios, can greatly facilitate data management, process control, and operational efficiency in continuous processing.
Conclusions
For any given therapeutic, biopharmaceutical development and manufacturing are handled by different parts of an organization, or even by different organizations altogether. Depending on the organization (size, organizations with all functional groups co-located, organizations with geographically separated functional groups, organizations that outsource various process stages), their operational needs can be very different, with little in common but the bioprocess between them.
The equipment and automation gaps that frequently exist between development and manufacturing can be effectively closed with the appropriate choice of bioprocess equipment. A modular equipment platform provides the highest degree of flexibility. Turnkey (measurement and control included), modular systems are preferred in environments where little or no automation infrastructure exists, a characteristic common in development environments. In manufacturing, where a preexisting automation infrastructure is more likely to be present, such modular systems can be integrated with or without their measurement and control subsystems. The availability of open communication standards further facilitates equipment integration within both SCADA-PLC and DCS environments.
Selecting an equipment supplier capable of providing multiple unit operations, each on modular product platforms, combines the benefits described above with single-source responsibility for integration and operation, documentation, and support. Suppliers with product offerings in single-use equipment, along with associated consumables, provide even more options for the configuration of effective bioprocess development and manufacturing. In the development stage, the up- and downstream systems can be scaled in concert, allowing physical interconnections and data management to be optimized. Moreover, transition to manufacturing scale will be faster and smoother. At the manufacturing stage, the various unit operations can be looked at as a more highly integrated bioprocess train.
Equipment choice ties the bioprocess to the automation architecture. Properly selecting an automation solution, especially one that spans the complete upstream-downstream bioprocess train under either the SCADA-PLC or DCS automation scenarios, can greatly facilitate data management and process control. With data connectivity and a comprehensive automation solution, alignment across all functions such as procurement, finance, production, material management, quality and more can be achieved, for an overall improved operational efficiency.
