Harvest & Collection
PRODUCTS
-
World's first real-time drinking water quality monitoring sensor for smart water meter. Online membrane integrity monitoring sensor for every vessel.
-
Whether you’re conducting an oncology trial, a neurology study, or imaging-based endpoint research, TrialKit’s flexible imaging capabilities can meet your needs.
-
Take advantage of our end-to-end DMPK expertise for optimizing compounds from discovery through IND filing for small and large molecules
-
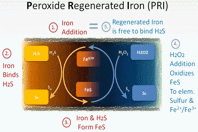
Sulfide odor control within sanitary sewers has been practiced for over 50 years, yet only recently have substantive advances been made. Where once the choice of chemical treatment was either chlorine or iron salts, safer and more environmentally benign technologies based on nitrates, hydrogen peroxide, and/or magnesium hydroxide have gained acceptance. These new alternatives, however, can increase treatment costs substantially and present limitations in themselves. Therefore, it makes sense to explore new avenues for lessening the adverse impacts of the older, cheaper mainstays, particularly iron salts (ferrous/ferric chloride or sulfate) that provide other benefits to wastewater treatment operations.
-
The MPBR-0122CSP3 is a compact, high-performance MMIC power divider/power splitter that operates across the 1 to 22 GHz frequency range.
WHITE PAPERS AND CASE STUDIES
-
Cooling Tower Crisis: How Hydrogen Peroxide Scrubbed Away The Bacteria Buildup
A West coast power plant needed an immediate solution to clean its cooling tower that was fast, cost effective, and did not require removal and manual cleaning of the tower's fill material.
-
Accelerating A Complex Molecule From CLD To cGMP In 12 Months
Learn how to mitigate technical risk, implement parallel process development, and design custom purification strategies for highly sensitive novel modalities to accelerate to cGMP material.
-
Vassar WWTP Eliminates Rags To Protect Equipment, Biosolids Quality
The Vassar wastewater treatment plant (WWTP) in Michigan confronted persistent challenges from non-dissolvable solids by adopting Duperon's Dual Auger System (DAS) to address these issues effectively.
-
Optimized Stability Storage
A healthcare manufacturer facing space constraints and regulatory pressures outsourced their stability storage needs to another company, resulting in reduced costs and focus on core competencies.
-
Data Key In Addressing Patient Recruitment Challenges
Explore how a Phase II trial for extensive-stage and previously treated small cell lung cancer overcame recruitment challenges with a data-driven solution designed to surface protocol-matched patients.
-
WuXiHigh 2.0 Delivers A 180 mg/mL Formula
This case study shows how smart formulation and integrated development achieved a stable 180 mg/mL biologic for subcutaneous delivery, for teams tackling concentration issues.
-
Navigate The Complexities Of CGTs With Regulatory And CMC Expertise
Explore how strategic regulatory and CMC expertise helped guide a biopharmaceutical company’s regulatory and development strategy for a cell and gene therapy product.
-
Electronic Batch Reporting
Learn how a CDMO automatically created a comprehensive batch report model to drive their reporting and speed root cause identification of quality failures with an Industrial DataOps software solution.
-
Vaccine Fill Line Operations
Learn how adopting a harmonized, risk-based validation strategy across global sites can reduce operational readiness timelines and drive improvements in equipment effectiveness and regulatory compliance
-
Enhancing Water Quality For Mountaire Farms
Mountaire Farms resolved their wastewater facility issues by replacing mechanical surface aerators with SDOX technology, saving $38 million and improving treatment.
-
How Fine Screens Simplify The New Waste Discharge Requirements
How are fine screen solutions aiding winemakers in their efforts to meet California State Water Resources Control Board mandates for enhanced monitoring and treatment of winery process water.
-
Working Smarter: Empowering Water Operators With Integrated Data
As water utilities face the challenges of sustainability, efficiency, and service quality, digital technology has become a necessity. The right tech can help deliver reliable service, optimize systems, and meet sustainability goals, but integrating the data streams these solutions generate can offer even greater gains – allowing utilities to move faster and achieve more powerful outcomes.
NEWS
-
Silo Pharma Expands Intellectual Property Portfolio With Patent Application For Exclusively Licensed Alzheimer's Drug4/28/2025
Silo Pharma, Inc. (Nasdaq: SILO) (“Silo” or the “Company”), a developmental stage biopharmaceutical company focused on novel therapeutics and drug delivery systems, today announced the filing of a patent application with the U.S. Patent and Trademark Office (USPTO) focused on the neurology drug SPC-14, an intranasal compound for the treatment of Alzheimer’s disease (AD) exclusively licensed to Silo Pharma from Columbia University.
-
U.S. Department Of Energy Announces $6.9M In Projects To Support Effective Community Waste-To-Energy Strategies For Local Transportation Needs1/7/2025
The U.S. Department of Energy's (DOE) Bioenergy Technologies Office (BETO) and Vehicle Technologies Office (VTO) announced $6.9M in funding for nine projects to support local waste-to-energy management solutions for transportation energy needs.
-
PNE Group Receives Permits For Three New Wind Farms In Germany7/8/2025
In the second quarter of 2025, the PNE Group received the necessary permits for the construction and operation of three onshore wind farms in the German states of Brandenburg, Hesse, and North Rhine-Westphalia.
-
Agilitas Energy Expands Into Hydropower With Acquisition Of Two Projects In West Virginia And Maryland6/24/2025
Agilitas Energy, a leading developer and operator of renewable energy and energy storage systems, today announced the acquisition of two late-stage hydropower development projects from Advanced Hydro Solutions.
-
Puraffinity Signs First Commercial Agreement For Full-Scale Deployment Of Its PFAS Removal Technology6/2/2025
The agreement was signed with Envytech Solutions, the leading expert in mobile water treatment in the Nordic region and a member of the Sortera Group, which operates across the Nordics and the UK.
ABOUT
Harvest and Collection
Harvest and collection is the process used to retrieve biological agents and vaccines present in cell cultures or to harvest and collect stem cells from blood or bone marrow. Many bioresearch companies have sophisticated systems for harvesting and collecting cells. These systems make counting cells easier and they also protect against contamination.
Cells are harvested once the cells reach a density level in the cell culture medium that precludes further growth. The best time to harvest cells is when they are in a confluent state (at least 50% of the culture dish is covered and before 100% of the dish is covered).
Cells can be harvested using on of three methods: Mechanical, using Proteolytic enzymes, or using EDTA. Mechanical harvesting uses a rubber spatula to remove the cells from the growth surface or culture. This method is quick but can cause many cells to die because it is highly disruptive. This method is favored when harvesting lots of different samples of cells to prepare extracts.
In this case, viability of the cells doesn’t matter. Three enzymes; Trypsin, Collagenase, and Pronase can be used in combination with EDTA. The combination of these enzymes with the EDTA makes cells detach from the growth medium.
This method is easy but also has a downside. It can damage the cell surface by eating up exposed cell surface proteins. EDTA can be used alone to detach cells from their medium and it is gentler than using trypsin.
Normally, collected cells are placed in a new suspension, or growth medium to continue growing new cultures. This is referred to as passaging or splitting the cells.