The Rise in Single-Use Instrumentation Options In Bioprocessing
By Brandon Haschke
As Single-Use manufacturing methods continue to take hold throughout the upstream and downstream biopharmaceutical sectors, improved instrumentation options are developing in parallel to meet the unique needs of Single-Use processes while reducing costs and maintaining performance.
Life sciences companies need operational flexibility because it helps them adapt to changing conditions and makes it easier to improve their processes. Oftentimes they may trade flexibility to gain better economies of scale, but they still want to keep their options open, often by using Single-Use technologies.
Traditional biopharmaceutical manufacturing methods are typically designed around stainless-steel equipment such as bioreactor tanks, piping and processing skids. These configurations can be built to any scale, from tabletop laboratory arrangements up to bulk manufacturing sizes. All the associated elements like instrumentation, valves and fittings have been designed for compatibility with these configurations. However, once stainless-steel systems are installed, their geometry constrains their operation, output and throughput to processes, requiring something similar to the original design.
The growing implementation of Single-Use manufacturing methods for upstream vessels and downstream processing equipment addresses these and other issues by providing options for scalability and operational savings, but it also introduces some new requirements. This is especially true with regards to how processes are instrumented and automated. Traditional automated measurement techniques are fixed-in-place just like the conventional equipment they are designed to be connected to, so the instruments and fittings must evolve for use with disposable Single-Use methods.
This article examines how Single-Use instrumentation options are developing and expanding for compatibility with this manufacturing method, while maintaining the familiarity and performance of traditional devices. Three of the most common pharmaceutical process measurements—pressure, pH and dissolved oxygen (DO)—are examined in detail.
How Single-Use Changes Manufacturing
Traditional installations of biopharmaceutical manufacturing processes rely heavily on fixed-asset stainless-steel technology (SST) for the vessels, equipment, piping, valves and pumps. These SST systems require significant up-front design and extensive on-site space for the primary equipment and supporting utilities.
Some of these SST elements are still necessary even if a manufacturer adopts certain Single-Use Technology (SUT) methods to supersede traditional equipment. However, there are many benefits to incorporating SUT methods with SST systems:
- Reduced capital installation costs
- Reduced operating costs
- Reduced chance of contamination
- Improved design flexibility and layout options
- Improved scalability
- Improved production planning
Capital costs for installing or upgrading SST systems include materials, fabrication and installation costs—all of which are largely avoided when using ready-made disposable pre-sterilized Single-Use bags and fittings.
Operating costs are lower for SUT portions of a processing system because comparable SST elements must undergo clean-in-place (CIP) and sterilize-in-place (SIP) procedures. These are expensive in terms of chemicals—including acid, caustic and other agents—and the associated handling and disposal. These processes are also energy-intensive with respect to utilities like steam and water-for-injection (WFI). Furthermore, CIP and SIP operations run the risk of impacting permanently installed traditional sensors in SST equipment. Because Single-Use bags and fittings are newly manufactured and sterilized before being put into service, these procedures are unnecessary, and cross-contamination from previous production is prevented.
Beyond these quantifiable improvements, SUT methods grant manufacturers far more flexibility than SST systems. Within the manufacturing suite the physical footprint is generally reduced, creating more layout options. Systems are more readily reconfigured to scale up or down, providing new opportunities to optimize production.
There are certainly some challenges to implementing SUT methods, particularly associated with how to instrument and automate them in a way comparable to SST systems, which will be addressed in more detail below. But in general, SUT offers possibilities and savings compared with SST systems.
Upstream and Downstream Can Benefit
Biopharmaceutical manufacturing processes are sometimes categorized as upstream, which includes production and harvesting, or downstream, where activities like pH adjustment, virus inactivation, filtration and bulk filling are performed (Figure 1).
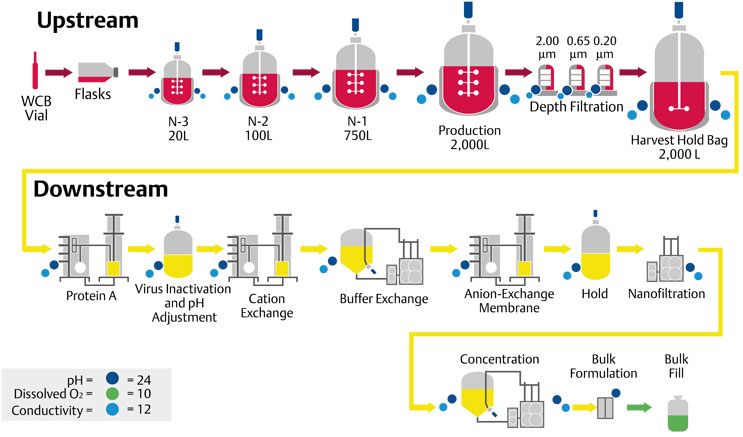
Figure 1: Process flow diagram (PFD) image from Emerson.
Single-Use pharmaceutical manufacturing techniques are available for upstream process vessels and downstream processing skids.
SUT methods have been primarily associated with bioreactor bags or holding bags (Figure 2), but the techniques can be applicable for many other upstream and downstream processes
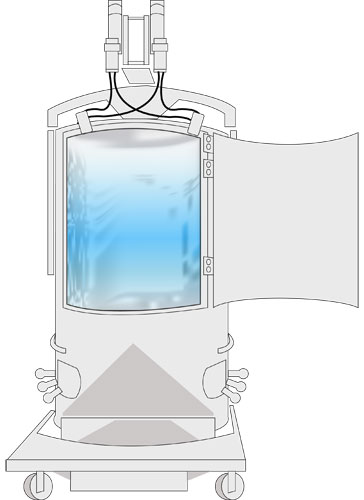
Figure 2: Image of a Single-Use bioreactor bag.
Only minimal stainless-steel infrastructure is required for Single-Use elements like this bioreactor bag, but instrumentation and fittings must be specialized for this service.
Single-Use bags are fabricated with pre-installed fittings and use materials compatible with the product and able to survive the gamma radiation sterilization procedure. Due to the requirement for product contact, pH sensors must be pre-installed in the bag fitting before the bag is sterilized.
It is also possible to install traditional DO and pressure sensors through standard fittings so they could contact the process, but this introduces difficulties for initial setup and calibration during operation. A more advantageous option is to use sensors based on traditional technologies but installed into specialized bag fittings that isolate the sensor from process product contact but still allow the measurement. In this way, the proven and reliable sensor electronics can be installed quickly as the bag is placed into service and even replaced during a production run or reused many times in other production runs.
For downstream use, Single-Use sensors and fittings can be installed directly into tubesets, depending on the geometry and form factor. This technique can be used to provide measurements in the transfer lines between production units. Because the Single-Use instrument market is growing, skid vendors can transition to support these methods with little difficulty.
Single-Use Instrument Requirements
To be suitable for Single-Use service, traditional instrumentation technologies must be upgraded to meet various requirements. This usually only applies to the sensor element because the instrument’s transmitter is generally installed remote from the sensor and connectorized for these applications.
Here is a summary of requirements that must be met for a sensor to be used in SUT service:
- Sensors pre-installed in Single-Use bags must survive gamma radiation sterilization
- Fitting connections must be leak-proof, aseptic and survive sterilization
- Sensor or fitting surfaces contacting product must meet wetted material requirements regulated by the FDA for leachable safety
- For sensors that can’t be removed once in service (pH), the installed sensor must be capable of being calibrated and tested before the equipment is put into service and the product is introduced
- For similar reasons, pH sensors must also remain serviceable, accurate and stable for the duration of needed use, commonly about 15 to 40 days for a batch
- For sensors that can be removed once in service (DO and pressure), the fitting must maintain the integrity and sterility of the process system at expected pressure, flow and temperature conditions
- Since SUT sensors may sit idle for long times in a bag or on the shelf, they must have a long shelf life, especially wetted sensors like pH
- Sensors must exhibit accuracy and stability comparable to traditional instruments
- Sensors and transmitters must be maintainable in similar ways to traditional instruments
- Sensors must be connectorized for easy interface with in-place transmitters
- Sensors and transmitters must integrate easily with automation systems
Meeting these goals is no small chore. Users want to maintain the performance of existing sensors, while gaining the advantage of SUT benefits. The following sections describe how Emerson’s Rosemount™ sensors meet these challenges for three types of process measurements.
Pressure for Single-Use Service
Pressure is the most straightforward process sensor that can be updated for SUT service. In fact, the sensor technology itself remains the same as for traditional sensors. The key is a disposable fitting manufactured into a Single-Use bag. This fitting features a flexible diaphragm which isolates the process fluid from the sensor element, while allowing the physical pressure to transfer to the sensor (Figure 3).

Figure 3: Closeup of Single-Use pressure sensor.
Pressure sensors for Single-Use are similar to traditional versions, but they are configured for installing into disposable fittings that isolate them from the process.
This combination allows the sensor to be reused, lowering the consumables cost since only the fittings change from one batch to the next. The removeable sensor is robust and highly accurate, and the fitting allows the sensors to be removed, replaced or recalibrated if required, even during a batch run.
Dissolved Oxygen for Single-Use Service
Much as for pressure sensors, DO sensors for Single-Use service use traditional measurement technology installed into an SUT-specific fitting. What is special about this fitting is that it features a permeable oxygen membrane so that the DO sensor has no direct contact with the process media but can measure the process value (Figure 4).

Figure 4: Closeup of Single-Use pH sensor.
The enhanced design of Emerson’s Rosemount 550pH Single-Use Sensor allows the sensor to be stored wetted, and then calibrated while in service at any time without removal.
This membrane does introduce a small lag time compared to direct contact, but this is not an issue for these types of applications. Furthermore, significant testing was performed by Rosemount to ensure the DO sensor would not need to be de-rated in any way for accuracy or linearity. This sensor can also be removed, replaced or recalibrated—even during production if needed. Calibrations performed between batches can reduce start-up time.
pH for Single-Use Service
Measuring pH in a Single-Use manner is more complicated than measuring pressure or DO. The glass electrode measurement technique requires the sensor to be in direct contact with the process media, which means these sensors by nature must be Single-Use and are subject to additional constraints. Since pH is a critical process parameter (CPP) for any bioreactor, users need sensors with zero drift and a long shelf life, so any concessions necessary for Single-Use service must not compromise those criteria.
Emerson has developed a sensor and fitting with a unique setup allowing the sensor to be stored for up to two years while wetted in a stable, proprietary buffer (Figure 5). The sensor ships retracted into the fitting, with O-rings used to keep the element wetted with a pharmaceutical grade buffer complying with USP <788>. This buffer is stable over the two-year shelf life of the sensor, and the known pH of the buffer can be used for calibration during start-up to ensure the sensor is operating properly. When ready for operation, the user simply pushes to insert the sensor element into the process media.

Figure 5: Closeup of DO sensor installed in adapter
This Rosemount 550DW Single-Use Dissolved Oxygen Sensor Adapter incorporates a gas permeable membrane that maintains process integrity and sterility under specified conditions, even if the sensor is removed in service for calibration.
Even with these modifications, the sensor achieves stability of less than 0.005 pH drift per day, enabling users to do a one-point calibration at the start of the batch and operate for a 20-day run after insertion without any required recalibration or maintenance under most conditions. This one-point calibration at start-up is highly accurate to the buffer pH, within 0.1 pH, so no other adjustments are required, leading to a long run time with no maintenance.
Instrumentation and Automation are Ready for Single-Use
Adoption of Single-Use manufacturing methods for biopharmaceutical processes is a growing trend due to the flexibility provided and the comparatively low capital cost commitment of SUT methods compared to SST systems. For best acceptance, SUT instrumentation and automation approaches must overcome the unique challenges of SUT installations while delivering similar performance. A new generation of innovative sensors are now available to address the issues by using specialized materials and form factors suitable designed for SUT service and providing instrument and processing functionality equal to traditional techniques.
About the Author
 Brandon Haschke is a product manager focusing on life sciences for Emerson's Rosemount instrumentation business. He is currently working on measurement devices to meet the needs of the Single-Use bioprocessing market and the unique challenges of this emerging manufacturing space. Brandon is an active member of BioPhorum and holds a degree in chemical engineering from the University of Minnesota.
Brandon Haschke is a product manager focusing on life sciences for Emerson's Rosemount instrumentation business. He is currently working on measurement devices to meet the needs of the Single-Use bioprocessing market and the unique challenges of this emerging manufacturing space. Brandon is an active member of BioPhorum and holds a degree in chemical engineering from the University of Minnesota.
All supplied figures are courtesy of Emerson
