Single-Use Connections Advance Aseptic Processing
By John Boehm, Colder Products Company

Today’s market demand on biologic and pharmaceutical manufacturers to develop new drugs — combined with the economic environment — is challenging bioprocessors to review their processing systems and seek ways to make them more flexible, reliable, and cost-effective. Increasingly, manufacturers are turning to single-use aseptic processing systems to meet or beat aggressive product-introduction time frames and control costs.
Innovative single-use technologies provide pharmaceutical manufacturers greater flexibility for replacing traditional stainless tubing, equipment or even entire process suites with plastic-based processing solutions. The benefits of converting to pre-sterilized single-use systems have been documented in numerous articles and case studies, and these benefits would be lost if manufacturers could not safely and securely connect the various pieces of equipment to create a complete aseptic process.
The connecting device or connection method may appear to be a small part of an overall system, however, connection and disconnection of tubing for process fluid transfer is a critical aspect of single-use processing. Manufacturers need to carefully consider the available options because the connector can be the deciding factor in keeping the single-use bioprocess truly aseptic.


STERILE CONNECTION OPTIONS
There are many ways by which biopharmaceutical manufacturers can create a sterile connection within or between their unit operations. The right selection is highly dependent on the needs and preferences of each facility.
Quick disconnect couplings or fittings (sanitary and luer) are commonly used with a variety of tubing types and sizes. These connectors provide quick, easy and secure connections when used in conjunction with laminar flow hoods. Tube welders are an alternative for creating sterile connections with thermoplastic elastomeric tubing of half-inch diameters and smaller. These systems use heated, replaceable blades to maintain sterility while welding two separate tubes together. Single-use Steam- In-Place (SIP) connectors create sterile connections between a variety of single-use systems and stainless processing equipment. These solutions require a steam supply at the point of connection. There are a number of single-use sterile connectors being introduced that allow a tubing-to-tubing sterile connection to be completed without the need for a laminar flow hood or tube welder. This capability opens up the option to create sterile fluid transfer between separate single-use systems even in gray space. In addition to sterile connectors, there is a sterile disconnect coupling with a shutoff valve that maintains the fluid sterility during or after disconnection of single-use tubing, even in an uncontrolled environment.
For increased flexibility, combination connectors with both a connect and disconnect function have been created, allowing for a sterile connect and disconnection throughout the process. For assemblies that do use an open format MPC connector, they can convert to a closed connection with a combination connector with an aseptic connector on one side and the connecting half to the MPC. For manufacturers that need a hybrid stainless steel and single-use processing equipment, AseptiQuik STC Connectors integrate the AseptiQuik sterile connector and the Steam-Thru II SIP connector.
Deciding which solution is best suited for a particular application depends on a number of factors such as fluid being processed, tubing selection, flow requirements, and space availability for laminar flow hood, SIP or tube welding equipment. Additional factors to consider include material compatibility, product availability, whether the connection method is already validated for use within the facility or organization, and supplier technical support.
BEST PRACTICE GUIDES
With the range of solutions available, it can be difficult for manufacturers to determine the best approach to evaluate or qualify connection technology for a particular process. The BioProcess Systems Alliance (BPSA) Technical Committee formed subcommittees to address best practices for major categories of single-use technologies including films and containers, filter capsules, tubing, and connectors and fittings. Because plastic connectors have different characteristics when compared with traditional stainless valves, a team of connection experts from various companies developed consensus guidelines on quality test methods used to address key performance attributes of single-use connection technology.
The result of this collaborative effort is a matrix that outlines tests common to connectors and fittings, test frequency, and a summary of available test references or industry standards. Tests include burst, integrity (leakage), bacterial challenge/soiling, biocompatibility, particulate, physicochemical, flow rate/ pressure drop, and sterilization process compatibility testing. This matrix is an excellent reference for drug manufacturers when developing an approach for selecting, qualifying and validating connection solutions. Representatives from BPSA member companies also can provide valuable support and assistance to users during technical evaluation.
APPLICATION EXAMPLES
Fermentation Seed Trains with Stainless Reactors:
Modern bioprocessing facilities often feature production bioreactors with capacities of 1,000 to 25,000 liters. Scaling up inoculum from a few million cells in several milliliters of culture to these production volumes is a challenge that requires an aseptic transfer at each point along the seed train. Traditional bioprocessing facilities accomplish scale-up using a dedicated series of stainless steel bioreactors linked together with valves and rigid piping. To prevent contamination between production runs, a Clean-In-Place (CIP) system is designed for each bioreactor, vessel, and piping line to remove any residual materials. Sterility assurance at the start of each culture is provided via an SIP system consisting of steam pipes, temperature sensors and condensate collection piping. These CIP and SIP systems require extensive validation testing and the valves and piping contained in these systems can create additional validation challenges. In addition, CIP and SIP systems must be revalidated following significant maintenance or changes to connecting piping and valve networks.
Figure 1: Fermentation seed train with stainless bioreactors
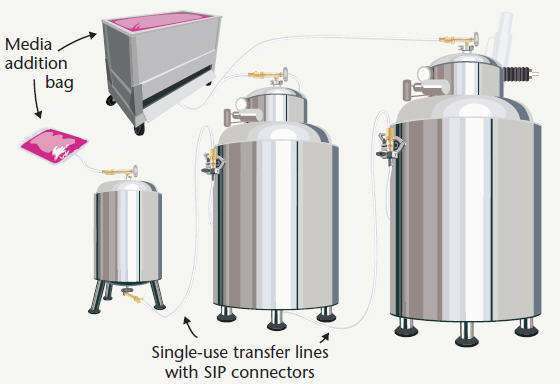
Advances in single-use system technology allow engineers to replace most storage vessels and fixed piping networks with single-use storage systems and transfer lines. Single-use eliminates the need for CIP validation for many components, reduces maintenance and capital expenses, and eliminates expensive vessels, valve, and sanitary piping assemblies. Figure 1 depicts a production suite that relies on stainless steel bioreactors, but integrates single-use technology for cell culture media storage and key transfer lines. Media storage systems arrive at the bioprocess facility pre-sterilized by gamma irradiation, often fitted with integrated filters, sampling systems, and connectors. Using a SIP connector allows operators to make sterile connections between these pre-sterilized single-use systems and stainless steel bioreactors for aseptic transfer of media.
Similarly, single-use transfer lines can transfer inoculum between bioreactors using either a peristaltic pump or headspace pressure. Such transfer lines reduce the number of reusable valves required for transfer, as well as eliminate problem areas for CIP and SIP validation. Terminating each pre-sterilized transfer line with a single-use SIP connector provides sterility assurance equal to that of traditional fixed piping, at lower capital costs.
Figure 2: Fermentation seed train with single-use bioreactors
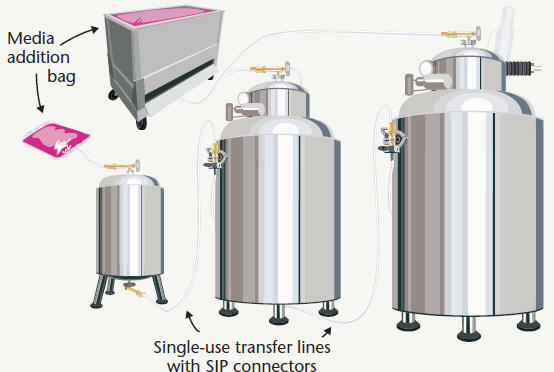
With the increased acceptance and availability of single-use bioreactor (SUB) technology, users can create flexible seed train systems consisting entirely of various volume SUBs or a combination of SUBs and traditional stainless bioreactors when high volumes are required. It is not practical to have a train of SUBs assembled, sterilized, and installed as one complete system so single-use connections can be used to provide a secure aseptic process by joining the various components together once they are installed within the process suite. As outlined above, this can be done with quick connectors, tube welding or sterile connection devices. In the event that smaller-volume SUBs are connected with larger-volume traditional stainless reactors, quick connects or sanitary fittings can be used in conjunction with laminar flow hoods, or SIP connectors can be used to establish sterile media transfer.
Figure 2 shows a model facility that incorporates single-use bioreactors in combination with stainless steel bioreactors for the two largest vessels.
Buffer/Media Application:
In-house media/buffer preparation requires inline sterile filtration after mixing and prior to storage. Many manufacturers are using filter integrity testing to ensure product purity and minimize product loss. Filter integrity testing verifies that the filters used are functioning properly and are capable of removing a minimum particle size from the filtrate. These tests can be conducted prefiltration, post-filtration, or both, to identify potential breaches in sterility and if a rerun of a batch process is necessary.
Historically bioprocessing facilities used hard-plumbed systems with replaceable filter elements in stainless steel filter housings, which required time-consuming setup, validation, and post-production cleaning. Introducing single-use components into these filter integrity test systems reduces the risk of contamination in addition to improving process flexibility. Single-use components used in filtration and filter integrity testing include disposable capsule filters, bags, tubing, clamps, and connectors. Connectors are a vitally important interface between the components within these filter systems and also provide quick and easy integration of the subsystem into the larger production process.
Figure 3: Buffer/media preparation
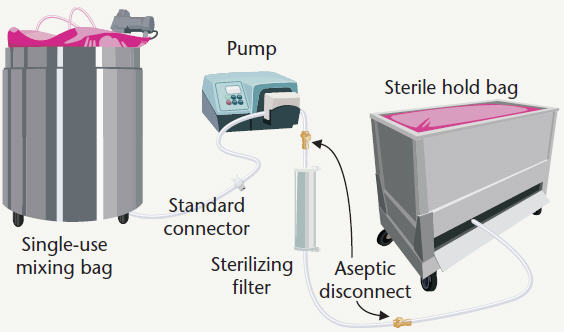
Single-use components and systems can replace some or all of the stainless steel equipment used for culture media and buffer preparation (Figure 3). Single-use tank liners or specialty mixing bag systems substitute for fixed-placed mixing tanks. Bag systems with integrated capsule filters replace both stainless filter housings and sterile hold tanks. After the process fluid has been filtered into the sterile hold bag, the filter can be quickly and easily detached for post-filtration integrity testing using valved sterile disconnect couplings. The resulting sterile disconnection enables the technicians to confidently remove the filter without the risk of contaminating the contents stored in the holding bag, while also keeping the filter wetted for integrity testing. After removal, filters may be tested using automated equipment by bubble-point, gas diffusion or pressure decay methods. Once testing confirms filter integrity, the stored cell culture media may be released for continued processing. Extending the use of single-use components to filter integrity testing is an additional way manufacturers can streamline the bioprocess and get the product to market faster.
Single-Use for Suite-To-Suite Transfer:
After the completion of the upstream production, the protein-containing medium must be aseptically transferred to a different location in the production facility to be prepared for the final filling operation. This suite could be in a room next door or much farther away. Traditional bioprocessing facilities accomplish this transfer by using stainless steel manifolds with piping or reusable hoses as transfer lines. This equipment requires validated CIP and/ or washing procedures prior to use and sterilization before each media transfer.
With the acceptance of single-use components, process engineers are incorporating single-use transfer lines between the bioreactor in the process suite and the transfer vessel to simplify this process. Using pre-sterilized connectors and tubing, the medium can be moved from the production suite to the preparation suite without the need to sterilize stainless steel piping or equipment. In addition to faster production, these single-use systems also give the manufacturer greater flexibility when determining which process to run in each production suite. The enhanced mobility eliminates many of the restrictions that hard-plumbed piping can place on the manufacturing facility. The risk of cross-contamination in the suite-to-suite transfer process can be high, especially in multi-product facilities; utilizing sterile connectors as an interface between key processes can reduce these risks and improve the speed and safety of drug development and delivery.
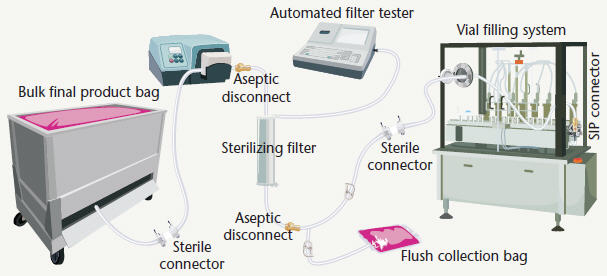
Figure 4: Final fill operation
Formulation and Fill:
Product safety concerns and overall product value are highest during final formulation and dose filling. Like the previous applications, traditional filling processes consist of stainless steel equipment connected via reusable valves, rigid tubing and steel pipes. Once again, single-use technology in the form of bulk storage containers, filters, tubing, connectors, and even disposable filling manifolds can be integrated to minimize contamination risks and reduce operational downtime.
Figure 4 depicts a filling operation utilizing a single-use manifold system with a sterile filter to transfer the drug product between the bulk storage bag and the filling equipment in the isolator. To address product quality and reduce waste, both pre- and post-filtration integrity testing may be combined during the final production stages.
To conduct pre-filtration testing, the filter assembly is connected to the sterile hold tank using a sterile connector. Once connected, the filter is wetted using product formulation and then pre-filtration testing can be performed. After filter integrity is confirmed, the line to the flush bag is clamped off and the filter assembly is aseptically connected to the isolator transfer line. The isolator line can be pre-installed to the vial filling system using an SIP connection minimizing downtime. The flow clamp is then opened for filtration to begin. Once filtration is complete, the filter can be removed from the assembly using sterile disconnect couplings to maintain the sterility of the filter until post-filtration integrity testing can be performed.
By conducting both pre- and post-filter integrity tests, operators can be assured that product purity has been maintained in the final drug formulation and that it is ready for release.
FUTURE GROWTH FOR SINGLE-USE CONNECTIONS
Connection and disconnection of tubing for fluid transfer is critical to aseptic processing with single-use systems or in combination with traditional stainless steel equipment. Sterile connection technology enables different subsystems or processes to be combined to create increased flexibility and efficiency from upstream fermentation through downstream filling. Because the connector can be the deciding factor in maintaining process sterility, users need to carefully select their connection solutions.
Industry support from organizations like the BPSA along with continuing product development from suppliers will further address aseptic processing needs of faster product changeover for small batch production. Larger throughput systems and tubing ranges will create even more efficient operations and give manufacturers the flexibility they need to get their products to market faster. As a result, the advancements in connection technology are helping address industry demands for rapid drug development, process reliability, and improved manufacturing efficiencies.
ABOUT JOHN BOEHM
John Boehm is the Vice President of Biopharma at CPC. John is the former Vice Chair of the BioProcessing Systems Alliance (BPSA) and has also served in the roles of Education Chair and Lead for the Connectors and Fittings Subcommittee. John has a bachelor’s degree in mechanical engineering and a master’s degree in business.
References:
1. “Complete BPSA Component Quality Test Matrices,” Author/s: BPSA Technology Committee, BioProcess International, November 2015.
2. “Bio-Process System Alliance Component Quality Test Matrices,” Author/s: BPSA Technical Committee, BioProcess International, April-May 2007.
About CPC
CPC (Colder Products Company) is the leader in single-use connection technology, offering a wide variety of connectors for biopharmaceutical manufacturing. Innovative, flexible designs easily combine multiple components. The company’s well-known AseptiQuik® and MicroCNX® connectors provide quick and easy sterile connections even in non-sterile environments. Learn more about these connectors and our entire portfolio at cpcworldwide.com/bio. Connect with confidence with CPC, an operating company within Dover Corporation.
