A Design Of Experiment (DOE) Approach For Purification Of Recombinant Proteins
By Xuemei M. He, Sherif Hanala, and Mark Snyder, Bio-Rad Laboratories, Inc.
Mixed-mode chromatography has become an important purification tool for downstream process developers, who are always seeking highly selective yet robust methods for the purification of recombinant proteins. Mixed-mode chromatography matrices are designed to present multiple interaction modes to resolve target proteins and impurities, entities that are themselves complex multimodal molecules. Under specific purification conditions, one or more such interaction modes may be involved in the binding or repulsion between the target protein and the chromatography media. Therefore, the behavior of a protein during purification by mixed-mode chromatography is often not predictable on the basis of its pI or amino acid sequence.
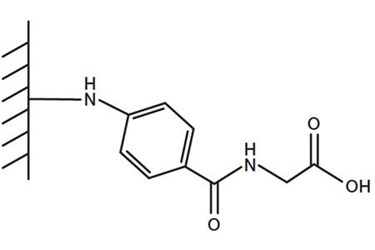
Here, we describe the use of a Design of Experiment (DOE) approach in the initial screening of chromatographic conditions for the purification of a diverse set of protein targets on Nuvia cPrime hydrophobic cation exchange media (Figure 1). The ligand on this media has three major functionalities: a weak carboxylic acid end group, an aromatic hydrophobic ring, and an amide bond serving as a potential hydrogen bond donor/acceptor. The combined effect of these structural elements provides unique selectivity and good conductivity tolerance, allowing protein purifications to be conducted effectively under gentle conditions. Our studies show that, with a limited amount of protein sample and chromatography media, a simple DOE setup can be used to determine the effects of buffer pH and conductivity on selectivity, recovery, and robustness of protein purification on Nuvia cPrime. Working conditions established by such scale-down studies can be used for the purification of a target protein on a preparative scale.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Bioprocess Online? Subscribe today.
