Pharmazyme Awarded SBIR Grant for Cytochrome P450 Platform
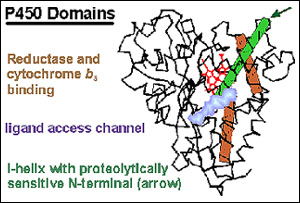
The cytochrome P450s are responsible for metabolizing a variety of important chemicals, such as steroids, fatty acids vitamins, pesticides, environmental and natural carcinogens, as well as therapeutic drugs. Hence, the ability to predict how individuals will metabolize drugs using cytochrome P450s will help determine drug toxicity, drug-drug interactions, and therapeutic dosing.
"We are pleased to be the recipient of this SBIR award," said ICC Vice President Steve DeGraw. "It is an endorsement of our company's emerging screening technology, particularly in the drug metabolism area."
Pharmazyme uses a novel system to develop assays that can detect single base polymorphisms in cytochrome P450 genes.
"With respect to cytochrome P450s and drug metabolism, we believe this system, along with our other products, will improve the safety of pharmaceuticals by both screening for patient populations with specific drug metabolism deficiencies, and by screening for drugs with unsatisfactory cytochrome P450 metabolism characteristics," said DeGraw.
Pharmazyme is a division of Immune Complex Corp., which specializes in the development of infectious disease vaccines. Pharmazyme, the company's most recent division, specializes in the development of drug metabolism products and services.
For more information: Steve DeGraw, Immune Complex Corp., 3347 Industrial Court, San Diego, CA 92121. Tel: 858-793-2661, x110. Fax: 858-793-2666. Email: sdegraw@pharmazyme.com.
