6 PEG Alternatives You Should Be Thinking About
By Alexander K. Aust, M.S., MBA, Aust Business Solutions
Nanoparticles (NP) are ultra-small particles typically ranging in size from 1 to 1,000 nanometers.1 NPs can be composed of various materials, including polymers, metals, lipids, and other materials. The composition and diminutive size of NPs establish unique properties and behaviors. In drug delivery, NPs serve as carriers for therapeutic agents, such as genetic material.1 Because of this characteristic, NPs play a pivotal role in improving the efficacy and safety of pharmaceutical treatments. Their small size grants them the ability to interact with biological systems on a cellular and molecular level, offering several advantages as a drug delivery system (DDS).
NP drug delivery optimization is a critical step in improving drug solubility and stability, particularly for drugs that exhibit poor water solubility or limited bioavailability or that elicit unwanted immunogenicity. NPs work by encapsulating and protecting these drugs from degradation in biological environments.1 This, in turn, enhances drug bioavailability and efficacy. Moreover, NPs can be engineered to release their cargo gradually, facilitating sustained drug release. Gradual drug release is particularly beneficial for medications requiring prolonged therapeutic effects. In addition, NPs can also be designed to target specific cells or tissues, minimizing off-target effects and reducing the required drug dosage, thereby lowering the risk of side effects.
Although NPs do exhibit many appealing characteristics as a drug delivery tool, additional modifications, specifically, molecule bioconjugation, may be required to minimize off-target effects, unintended immune response, and general toxicity.2 Polyethylene glycol (PEG) bioconjugation is most commonly used to resolve these issues. The conjugation of PEG to drug delivery carriers is a widely adopted strategy to enhance the therapeutic potential of drug carriers by promoting better pharmacokinetics, prolonged circulation in the blood, and improved drug delivery precision.2 PEG bioconjugation optimizes NPs by acting as a stealth polymer that prevents various bloodstream interactions and extends carrier lifespan in the body.2 Although PEGylation ultimately allows for more efficient drug delivery, prolonged circulation in the blood can ultimately trigger the body's immune response. This can lead to anti-PEG antibody production and, in turn, potentially life-threatening side effects like cardiopulmonary stress from anaphylaxis induction. For this reason, PEG alternatives have been heavily researched in order to combat possible toxicity. Below is a list of PEG alternatives currently being studied in the drug delivery field.
Polysarcosine (pSar)
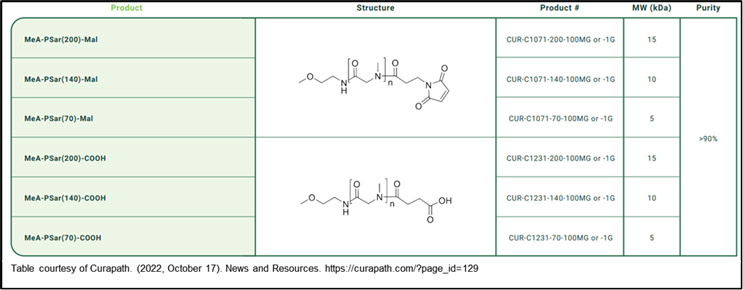
pSar bioconjugation involves attaching pSar chains to therapeutic molecules, such as proteins or nanoparticles.3 pSar is a biocompatible, synthetic hydrophilic polymer composed of repeating sarcosine units. Its unique structure allows it to interact with biological molecules while minimizing immunogenicity and clearance by the body's immune system. This modification can improve the drug's pharmacokinetics and biocompatibility, as PSARylation exhibits excellent water solubility and biostability. Its high-water solubility and low toxicity, combined with the ability to reduce the clearance of drugs from the bloodstream, makes PSARlyation a promising option to improve the therapeutic efficacy of various compounds.3 pSar bioconjugation is gaining traction as a valuable strategy to overcome the limitations of PEGylation, and it holds great promise in the development of advanced drug delivery systems. Some available pSar polymer conjugates on the market (shown in the Table) include Curapath’s monofunctional MeA-PSar(200)-Mal and MeA-PSar(200)-COOH and bifunctional Azide-PSar(200)-COOH and Azide-PSar(200)-NHS.4
Hydrophilic Polymers
Hydrophilic polymers offer a promising alternative to PEG for various biomedical applications. Hydrophilic polymers are short, hyperbranched polymers that are known to be biocompatible and non-toxic and elicit low immunogenicity. These polymers possess a high affinity for water, enabling them to enhance the solubility and stability of therapeutic compounds comparable to PEG.5 What sets hydrophilic polymers apart is their structural diversity; they come in various branched forms with unique properties, allowing researchers to tailor their selection to specific requirements. Moreover, many hydrophilic polymers, such as polysaccharides and polyvinylpyrrolidone (PVP), are more biocompatible and biodegradable than PEG, reducing concerns about potential immunogenicity and long-term accumulation in the body. These characteristics make hydrophilic polymers a versatile and effective choice for bioconjugation and drug delivery.
Zwitterionic Polymers
Zwitterionic polymers are a class of synthetic polymers that contain both positive and negative ionic groups within their molecular structure, which result in a net-neutral charge.5 These polymers possess a remarkable ability to mimic the behavior of natural biomolecules, making them highly attractive for biomedical applications. Zwitterionic polymers have gained attention as a potential replacement for PEG. These polymers are exceptionally hydrophilic, promoting water solubility and preventing protein fouling, which can occur when foreign substances adhere to the surface of medical devices or drug carriers.5 Additionally, their charge neutrality reduces the risk of immunogenic reactions compared to PEG, making them a more biocompatible choice for various applications in drug delivery.
Poly(amino acid) Based Lipopolymers
Synthetic poly(amino acid) (PAA)-based liposomes represent an innovative approach in the realm of drug delivery systems. These liposomes are encapsulated within a protective layer composed of synthetic poly(amino acids), which serve as stealth agents to evade the body's immune system.6 Unlike PEGylation, PAAs offer a more tailored and biocompatible solution. The customizable nature of PAAs allows for precise control over the size, surface charge, and stability of the liposomes.6 Additionally, PAAs exhibit excellent biodegradability and biocompatibility, minimizing concerns related to long-term accumulation and potential immunogenic responses. Some examples of PAAs include polyglutamic acid (PGA), poly(hydroxyethyl-l-asparagine) (PHEA), and poly(hydroxyethyl-l-glutamine) (PHEG). PAA liposomes have shown promising results in enhancing the circulation time of drugs in the bloodstream and improving targeted drug delivery to specific cells or tissues.7 As a result of these benefits, PAA-based stealth liposomes have emerged as a viable alternative to PEG.
XTENylation
XTEN is a biopolymer engineered from a repeating amino acid sequence with high structural homogeneity.8 XTENylation can be precisely tailored to modulate the size, charge, and hydrophilicity of therapeutic molecules or polymers.8 XTENylation has several advantages over PEGylation such as reduced immunogenicity, as it is made from naturally occurring amino acids and is less likely to trigger an immune response. XTEN polymers are also highly biodegradable, thus addressing concerns related to long-term accumulation in the body.8 Furthermore, XTEN's modular design allows for specific customization, making it an attractive choice to improve the solubility, stability, and circulation time of therapeutic agents, while overcoming some of the limitations associated with PEGylation.
Poly(thioglycidyl glycerol)
Poly(thioglycidyl glycerol) (PTTG) is a synthetic polymer analogous to the plasma protein, α2-macroglobulin.9 PTTG’s synthetic hydrophilic methionine-mimetic structure offers versatile properties such as stealth from immune response, combating the accelerated blood clearance effect, and acting as an antioxidant.9 For these reasons, PTTG has the potential to replace PEG because of increased biocompatibility and the potential to enhance drug delivery and bioavailability. Although research on PTTG is relatively new, there is much promise for PTTG as a customizable alternative for enhancing the therapeutic efficacy of various pharmaceutical and medical devices in addition to addressing some of the challenges associated with PEGylation.
In summary, NPs have revolutionized drug delivery by offering unique advantages in improving the solubility, stability, and targeted release of therapeutic agents. While NPs hold great promise, concerns such as immunogenicity, off-target effects, and toxicity necessitate the development of alternative strategies to replace PEG bioconjugation. Several exciting alternatives have emerged, each with its own set of advantages.
These alternatives are reshaping the field of drug delivery, offering safer and more effective options to enhance the therapeutic efficacy of various pharmaceutical and medical devices while addressing the limitations associated with PEGylation. As research in this area continues to expand, the future of drug delivery holds great promise.
References
- Khan, I., Saeed, K., & Khan, I. (2019). Nanoparticles: Properties, applications and toxicities. Arabian Journal of Chemistry, 2019 November; 12(7), 908–931. https://doi.org/10.1016/j.arabjc.2017.05.011.
- Suk JS, Xu Q, Kim N, Hanes J, Ensign LM. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv Drug Deliv Rev. 2016 Apr 1;99(Pt A):28-51. doi: 10.1016/j.addr.2015.09.012. Epub 2015 Oct 9. PMID: 26456916; PMCID: PMC4798869.
- Yali Hu, Yingqin Hou, Hao Wang, and Hua Lu. Bioconjugate Chemistry. 2018 29 (7), 2232-2238. DOI: 10.1021/acs.bioconjchem.8b00237.
- Curapath. (2022, October 17). News and Resources. https://curapath.com/?page_id=129
- Hoang Thi TT, Pilkington EH, Nguyen DH, Lee JS, Park KD, Truong NP. The Importance of Poly(ethylene glycol) Alternatives for Overcoming PEG Immunogenicity in Drug Delivery and Bioconjugation. Polymers (Basel). 2020 Feb 2;12(2):298. doi: 10.3390/polym12020298. PMID: 32024289; PMCID: PMC7077443.
- Thompson M, Scholz C. Highly Branched Polymers Based on Poly(amino acid)s for Biomedical Application. Nanomaterials (Basel). 2021 Apr 26;11(5):1119. doi: 10.3390/nano11051119. PMID: 33925961; PMCID: PMC8145254.
- Metselaar JM, Bruin P, de Boer LW, de Vringer T, Snel C, Oussoren C, Wauben MH, Crommelin DJ, Storm G, Hennink WE. A novel family of L-amino acid-based biodegradable polymer-lipid conjugates for the development of long-circulating liposomes with effective drug-targeting capacity. Bioconjug Chem. 2003 Nov-Dec;14(6):1156-64. doi: 10.1021/bc0340363. PMID: 14624629.
- Haeckel A, Appler F, Ariza de Schellenberger A, Schellenberger E. XTEN as Biological Alternative to PEGylation Allows Complete Expression of a Protease-Activatable Killin-Based Cytostatic. PLoS One. 2016 Jun 13;11(6):e0157193. doi: 10.1371/journal.pone.0157193. PMID: 27295081; PMCID: PMC4905650.
- Richard d’Arcy, Farah El Mohtadi, Nora Francini, Carlisle R. DeJulius, Hyunmoon Back, Arianna Gennari, Mike Geven, Maria Lopez-Cavestany, Zulfiye Yesim Turhan, Fang Yu, Jong Bong Lee, Michael R. King, Leonid Kagan, Craig L. Duvall, and Nicola Tirelli. Journal of the American Chemical Society. 2022 144 (46), 21304-21317. doi: 10.1021/jacs.2c09232
About the Author:
 Alexander Aust, M.S., MBA, is the cofounder and owner of Aust Business Solutions, a pharmaceutical consultancy based in Maryville, TN, where he focuses on strategic planning and partnerships and technical support. The firm provides CMC and project management services, assists with regulatory submissions, and helps to implement new technology. Aust serves as a senior project manager for the Parenteral Drug Association. He earned his MBA from Ball State University and an M.S. degree from Purdue University in molecular and cellular biology.
Alexander Aust, M.S., MBA, is the cofounder and owner of Aust Business Solutions, a pharmaceutical consultancy based in Maryville, TN, where he focuses on strategic planning and partnerships and technical support. The firm provides CMC and project management services, assists with regulatory submissions, and helps to implement new technology. Aust serves as a senior project manager for the Parenteral Drug Association. He earned his MBA from Ball State University and an M.S. degree from Purdue University in molecular and cellular biology.
