News from ASCB: Centrosomes may provide clues to cancer prognosis
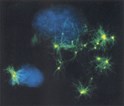
In the first study, Stephen Doxsey of the University of Massachusetts Medical School performed immunohistochemical studies using antibodies against the centrosome protein percentrin in pre-invasive cancers of the prostate, cervix and breast. These experiments showed that centrosome abnormalities exist prior to the onset of genetic instability and to the cytological changes indicative of tumorigenesis. In addition, inducing centrosome changes by over-expressing pericentrin led to chromosome abnormalities and cell defects that resembled those seen in aggressive tumors.
Furthermore, Doxsey and co-workers report that centrosome defects increase during prostate tumor progression and correlate with increased genetic instability. Quantitative studies of pericentrin in tumor tissues demonstrated that pericentrin levels were 2-4-fold higher in the cytoplasm and within centrosomes of tumor versus adjacent nontumor cells. Taken together, these results suggest that centrosome defects can cause genetic and cellular changes that occur during tumor progression.
Evidence for a Direct Role for Centrosome Defects in Human Tumorigenesis
Stephen Doxsey, Program in Molecular Medicine, University of Massachusetts Medical School, 373 Plantation St, Worcester, MA 01605.
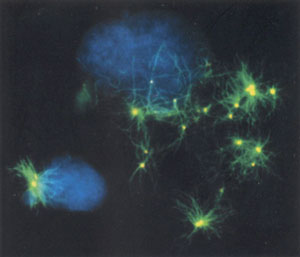
In the second study, Jeffrey Salisbruy of the Mayo Clinic Foundation in Minnesota examined the centrosome's role in hormone-dependent and hormone-independent breast cancers, and found that centrosome abnormalities mark the transition from hormone dependence to independence, an event that signals a change to a more aggressive cancer. Looking at the estrogen receptor positive cell line (MCF-7) and estrogen receptor negative cell line (MDA-MB 231 and MDA-MB 435), the researchers characterized the relationship between centrosome amplification and metastatic phenotype. They found that the near diploid MCF-7 cell line showed centrosomes of normal number and size while centriole amplification and aberrant mitoses were observed in the aneuploid MDA-MB 231 and MDA-MB 435 cell lines.
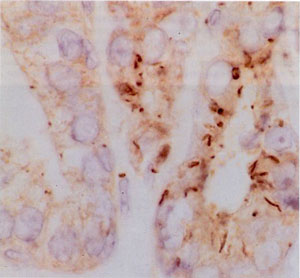
Furthermore, dissociating DNA replication or cytokinesis from centrosome duplication resulted in a marked increase of centrosome number in estrogen-receptor negative cell lines (MDA-MB 231 and more markedly in MDA-MB 435) while MCF-7 cells did not undergo centrosome amplification. These observations show that centrosome amplification is related to estrogen receptor status, DNA ploidy, and metastatic potential in breast cancer cell lines.
Centrosome Amplification And Genomic Instability In Hormone Dependent And Independent Breast Cancer Cell Lines
Jeffrey L. Salisbury, Tumor Biology Program, Mayo Clinic, 200 First St SW, Rochester, MN 55905.
