How Droplet Digital PCR Has Revolutionized Noninvasive Plasma-Based Detection Of Mutations

Research Background
MolecularMD is a preferred provider of molecular diagnostic products and services to pharmaceutical and biotechnology drug developers. Our capabilities range from specialty molecular testing performed in our centralized CLIA-certified and CAP-accredited laboratories to development of FDA-approved companion diagnostics.
Application
There is an unmet need for assays that monitor and detect the emergence of resistance mutations to targeted therapies, especially for advanced solid tumors where tissue availability is limiting. MolecularMD was interested in developing a plasmabased assay to detect EGFR T790M mutation in circulating tumor DNA as a method for monitoring the emergence of resistance to first-generation EGFR tyrosine kinase inhibitors. The level and amount of EGFR T790M, as opposed to simply the presence or absence of mutations, appear to be critical for successful therapy selection. Droplet Digital PCR (ddPCR™) enables quantification of EGFR T790M resistance mutations in circulating tumor DNA without the need for preamplification. In addition, absolute quantification is possible without the need for standard curves, which is especially useful in clinical laboratory testing.
Click Here to Learn More About Targeted Gene Editing
ddPCR Results
MolecularMD has successfully developed a ddPCR assay for detection of EGFR T790M resistance mutations in circulating tumor DNA. This assay has a sensitivity range of 1–2% with a minimal input of approximately 2 ng DNA.
The results in Figure 1 show concordance between ddPCR and allele-specific (AS)–PCR for a plasma sample from a patient positive for EGFR T790M. No mutant was detected in the control plasma sample for wild-type EGFR (as shown in Figure 1, A–C). In the patient plasma sample, FAM-positive droplets indicated the presence of the mutant EGFR T790M (as shown in Figure 1, D–F). Only ddPCR can quantify the ratio of EGFR T790M vs. total EGFR, which was 27.2% for this sample.
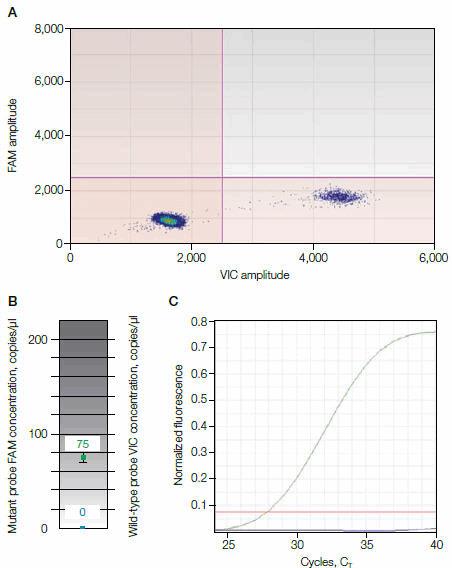
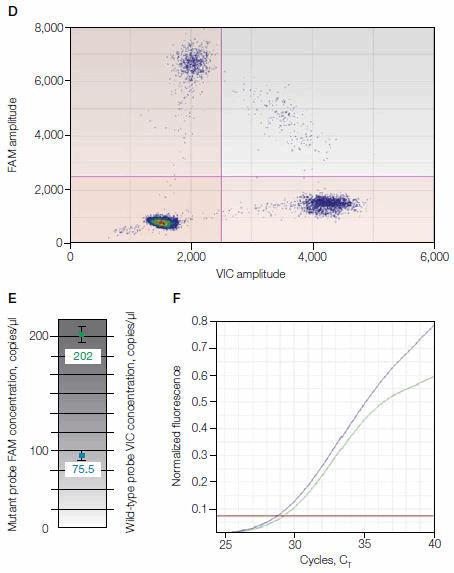
Fig. 1. Comparison of wild-type versus EGFR T790M detection in patient plasma. A–C, control plasma sample for wild-type EGFR (no mutant detected); D–F, plasma sample from a patient positive for EGFR T790M. CT, threshold cycle.
Conclusions
The ddPCR method allows detection of EGFR T790M mutations in circulating tumor DNA in the presence of abundant wild-type DNA with minimal input of sample DNA, which is especially relevant for circulating tumor DNA.
“Droplet Digital PCR offers a much better alternative for detection of rare mutations and copy number analysis.”
References
Kristof J et al. Absolute quantification of EGFR activation and resistance mutations as well as copy number in circulating nucleic acids by Droplet Digital PCR [unpublished data]. Poster presented at: American Association for Cancer Research Annual Meeting; April 6–10, 2013; Washington, DC.
Kristof J et al. (2012). Non-invasive absolute quantification of EGFR activating mutation L858R and gatekeeper mutation T790M in NSCLC plasma samples using Droplet Digital PCR. Eur J Cancer 48, S139–S140. FAM and VIC are trademarks of Applera Corporation.
