Extracellular Secretion And Non-Canonical Amino Acid (ncAA) Incorporation: New Capabilities Supporting Conjugate Vaccine Discovery And Development
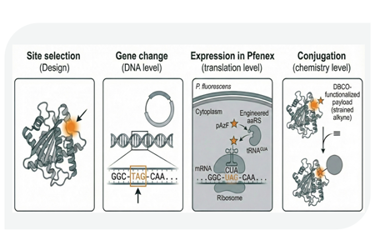
Conjugate vaccine development faces persistent challenges: complex purification processes that increase costs, inconsistent protein folding that compromises quality, and limited control over conjugation sites that reduces efficacy. Traditional expression systems often force developers to choose between yield and quality, while purification requires extensive cell lysis steps that introduce contaminants and drive up manufacturing costs.
A Pseudomonas fluorescens-based platform addresses these bottlenecks through two complementary approaches. Engineered host strains enable extracellular secretion, moving properly folded proteins directly into culture medium where they can be recovered without cell lysis, eliminating major purification steps while reducing host cell protein, DNA, and endotoxin contamination. Simultaneously, the platform achieves high-fidelity non-canonical amino acid incorporation at titers up to 15 g/L with 100% incorporation efficiency, enabling precise, site-specific bioconjugation. Hydrogen-deuterium exchange analysis identifies optimal conjugation sites on carrier proteins like CRM197, while maintaining expression titers comparable to native protein.
Access the poster below to examine the data, intact mass analysis confirming incorporation fidelity, and comparative results across multiple vaccine-relevant antigens, including VLPs, membrane proteins, and toxoid constructs.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Bioprocess Online? Subscribe today.
