Development Of Non-Affinity Based Purification Platform For Neutral/Basic IgMs
By J. Greenwood II, W. Rushton, P. Khandelwal, and C. Voss
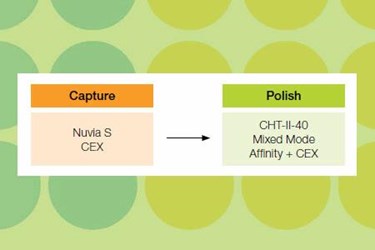
The biological properties of IgM antibodies make them very effective vehicles for in vitro diagnostics and therapeutics. However, purification of IgM antibodies is far more difficult than that for the more common IgG antibodies due to their complex structural and biochemical characteristics. Furthermore, the commonly available affinity chromatography supports (Protein A and Protein G) have little or no binding capacity for IgM.
Here we propose a non–affinity based two-column platform protocol for IgM purification. The strategy utilizes the cation exchange (CEX) resin, Nuvia™ S, for capture and the mixed-mode media, CHT™ Ceramic Hydroxyapatite, for polish purification. This workflow is suitable for purification of neutral and basic IgMs. We optimized the protocol with three different IgMs and present reducing and nonreducing PAGE images of the purified samples. This strategy is simple, scalable, and efficient and thus well suited for purification of diagnostic IgMs.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Bioprocess Online? Subscribe today.
